 
Three terminal atoms (no lone pair electrons) Two terminal atoms (ONE lone pair electrons)įour electron groups around the central atom: Tetrahedral geometry bond angle = 109.5° Examples: CF4, SiCl4, CH2Cl2, CHCl3 Tro: Chemistry: A Molecular Approach, 2/eġ5 Tetrahedral Geometry Tro: Chemistry: A Molecular Approach, 2/eġ6 Non-Octet: Trigonal Bipyramidal Electron Geometryĥ electron groups around the central atom: Trigonal Bipyramidal geometry Axial positions are above and below the central atom Equatorial positions are in the same base plane as the central atom LP-BP > BP-BP Stronger repulsive force from LP _ angles from LP _ BP-BP angle Tro: Chemistry: A Molecular Approach, 2/eīP electrons are shared by two atoms Negative charge in BP partially “neutralized” by nuclear charge less repulsive LP electrons are localized on the central atom negative charge from LP takes more space Tro: Chemistry: A Molecular Approach, 2/e Three electron groups around the central atom: electron groups taking a trigonal planar geometry bond angle = 120° Examples: COCl2, CH2O, SO3, BF3 Tro: Chemistry: A Molecular Approach, 2/eġ2 Trigonal Geometry Tro: Chemistry: A Molecular Approach, 2/eġ3 3D Animation: Three Electron Groups on Central Atom (bond angle _) Two electron groups around the central atom: electron groups occupy positions on opposite sides of the central atom Linear geometry Bond angle = 180° Tro: Chemistry: A Molecular Approach, 2/e #Electron groups on central atom = 2, 3, 4, 5, 6 5 basic arrangements of electron groups around a central atom Rarely >6, like IF7 e-group arrangements leads to different geometries Lone pair causes slight different geometry than bonding pair Resonance does NOT affect the electron geometry Tro: Chemistry: A Molecular Approach, 2/e 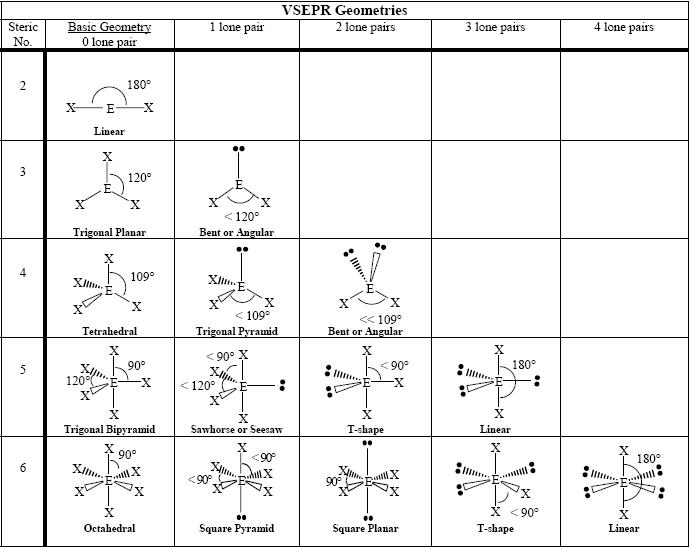  
_ electron groups on N Tro: Chemistry: A Molecular Approach, 2/e.VSEPR groups: Regions of Valence Electrons Each lone pair of electrons constitutes one electron group on a central atom Each bonding region constitutes one electron group on a central atom regardless of whether it is single, double, or triple O N VSEPR Theory Electron groups around the central atom repel each other (same “-” charge) Electron groups are spaced away to minimize the repulsion –valence shell electron pair repulsion theory (VSEPR theory) VSEPR theory can predict the shapes and bond angles in the molecule Tro: Chemistry: A Molecular Approach, 2/e.Lewis theory predicts there are regions of electrons in an atom Electron groups (= VSEPR group): Bonding pairs and Lone pairs Bonding pairs: Shared pairs of valence electrons between bonding nuclei Nonbonding pairs (= Lone Pairs): Unshared valence electrons on a single nuclei Tro: Chemistry: A Molecular Approach, 2/e Molecular Geometry: Shape of a molecule Two important factors in Molecular Geometry: Bond Angle Bond Length Tro: Chemistry: A Molecular Approach, 2/e Properties of molecular substances depend on the structure of the molecule The structure includes many factors, such as: the skeletal arrangement of the atoms the kind of bonding between the atoms ionic, polar covalent, or covalent the shape of the molecule Bonding theory should allow you to predict the shapes of molecules Tro: Chemistry: A Molecular Approach, 2/eĥ Molecular Geometry Molecules: 3-dimensional objects Sugar molecules (“Key”) fit into the active site of taste cell receptors (“Lock”): When the sugar molecule enters the active site, parts of the taste cell receptor split apart ion channels in the cell membrane to open resulting in nerve signal transmission Artificial sweeteners also fit into the same receptor, sometimes binding even stronger than sugar (making them “sweeter” than sugar) Tro: Chemistry: A Molecular Approach, 2/e Presentation on theme: "VSEPR theory Molecular Polarity"- Presentation transcript:Ģ Taste The taste of a food: Interaction between food molecules and taste cells Factors: Shape of the molecule and charge distribution within the molecule Food or “spicy” molecule fit snugly into the active site of specialized proteins on the surface of taste cells When this happens, changes in the protein structure cause a nerve signal Tro: Chemistry: A Molecular Approach, 2/e 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
